2 Penny Stocks “Strong Buy” Could Rise to $15


Which stocks are fan favorites or must be avoided? penny stocks. These stocks under $5 each are particularly divisive on Wall Street, with supporters and opponents making strong arguments.
These names are too tempting for risk-taking investors to ignore. With a low price, you get more money. On top of that, even a small increase in a stock’s price can translate into a large percentage gain, and thus, large returns for investors.
However, there is a but here. Critics point out that there could be reasons for the bargain, whether it’s poor fundamentals or too strong headwinds.
Based on the above, removing long-term underperforming stocks from penny stocks that are turning to gold can pose a significant challenge.
To assist with the due diligence process, we used TipRanks database only enter penny stocks that have received bullish support from the analyst community. We’ve found two stocks backed by enough analysts to earn a “Strong Buy” consensus rating. Not to mention each offer has huge upside potential, as some analysts predict they will rise to $15 per share in the coming months.
Capricorn Therapy (CAPR)
The first stock to come under scrutiny is Capricor Therapeutics, a clinical-stage biopharmaceutical company working on new treatments for Duchenne muscular dystrophy (DMD), a physically developmental disease. Rare, genetic basis usually appears in boys around 4 years of age. and patients may lose the ability to walk by age 12. Capricorns are taking a cell- and exosome-based approach to developing therapeutic agents aimed at treating or even preventing cases of the condition. Serious DMD.
Capricor’s leading drug candidate is CAP-1002, a therapeutic agent based on cardiac stem cells (CDC), a proprietary technology of the company. The company’s CDCs have conducted numerous studies, including several early-stage human trials, since 2007 and about 200 DMD patients have participated. The drug candidate uses cardiac cell therapy for its proven immunomodulatory activity and trials of CAP-1002 are investigating its ability to encourage cell regeneration.
In recent months, Capricor has presented important results from the HOPE-2 open-label extension study of CAP-1002 in the treatment of patients with end-stage DMD. The results were considered positive and resulted in statistically significant improvements in musculoskeletal function, observable by an objective improvement in upper extremity function. The company is currently enrolling patients in study HOPE-3, a pivotal Phase 3 trial in a double-blind placebo-controlled model. Capricorn plans to release an interim data analysis of the HOPE-3 study in mid-2023.
Earlier this year, Capricor licensed the CAP-1002 in the United States to Nippon Shinyaku for a $30 million upfront fee. In addition, the company enjoys significant payouts of up to $705 million and double-digit royalties on sales in the United States.
The company is also engaged in ongoing discussions with the FDA regarding the completion and filing of the Biological Authorization Application (BLA) for CAP-1002. Acceptance of the BLA will be an important milestone towards the further commercialization of the product.
Cover this stock for Ladenburg Thalmann, analyst Aydin Huseynov expressed confidence in the company’s ability to leverage the potential of the CAP-1002. He wrote, “CAPR is continuing to enroll DMD patients in the HOPE-3 rotation… In the meantime, CAPR also appears to be looking for outsourcing opportunities for CAP-1002 in former territories. of the United States, which could lead to meaningful results. prepaid fees. It seems CAPR wants to build on a royalty/milestone model that some other platform companies have built, to focus on additional potential vaccines/treatments using using its proprietary exosome technology.”
“To us, it looks like 2023 will be a deal-making year for CAPR, which could strengthen CAPR’s balance sheet, strengthen confidence in CAPR’s exosome platform, and possibly strengthen CAPR’s exosome platform.” allowing the company to raise more equity capital for other exploration projects… Our price target for CAPR shows potential for a 2-3x increase in the event of a positive mid-cap CAP-1002 index. 2023 and/or external licensing agreement for the former US CAP-1002. Q4 2022.”
Indeed, Huseynov rates CAPR stock as Buy, and his $15 price target suggests strong upside potential of 275% over the next year. (To see Huseynov’s achievements, click here)
Huseynov isn’t the only analyst noticing a strong uptrend here; all three recent reviews of this stock have been positive, with a Strong Buy consensus rating. The stock is trading for $4 and its median price target is $15, matching Huseynov’s price with a 275% upside potential in one year. (View CAPR stock forecast on TipRanks)
Biopharmaceuticals Vor (VOR)
The second cheapest stock we’ll be looking at, Vor Biopharma, is another clinical stage medical research company. Vor is focused on developing new treatments for blood cancers that will ‘change the standard of care’ for these difficult-to-treat conditions. The company is using hematopoietic stem cells that have been engineered to enable precisely targeted cancer therapies for post-transplant use. The company’s goal is to protect healthy cells while exposing cancer cells to pharmaceutical agents.
Vor currently has two main drug candidates, VOR33 and VCAR33, in multiple research phases, including 4 preclinical phases, 1 clinical phase and 1 preparation phase for clinical trials. VOR33, the leading candidate, is an eHSC product, generated by genetically modifying a donor healthy HSC to knock out the CD33 surface target. The goal is to induce HSCs that are protected from post-transplant therapeutic agents, exposing cancer cells to targeted therapy. This candidate is the subject of a Phase 1/2 clinical trial, with initial clinical data to be released before the end of the year. The trial has nine sites that are currently active and are continuing to recruit patients.
VCAR33, the other leading candidate, is a CD33-directed mosaic antigen receptor T (CAR-T) agent that is being investigated for the treatment of adults with acute myeloid leukemia. (AML). The drug candidate is scheduled to be the target of a Phase 1/2 clinical trial next year, and the IND application is being rolled out in 1H23.
In a review of Vor for JMP, analyst Silvan Tuerkcan writes: “VOR33 data is still expected in Q4 of 22… We remain confident in this readout based on preclinical work from several groups showing successful generation of edited CD33KO eHSCs and natural history data in humans with spontaneous mutations leading to CD33 deficiency in HSCs We also have confidence in the cellular process, as VOR’s CSO, Dr. Tirtha Chakraborty, was involved in the successful translation. another engineered HSC for human engraftment (CTX001)… A successful engraftment would be significant, in our view, validating an important part of the VOR approach.”
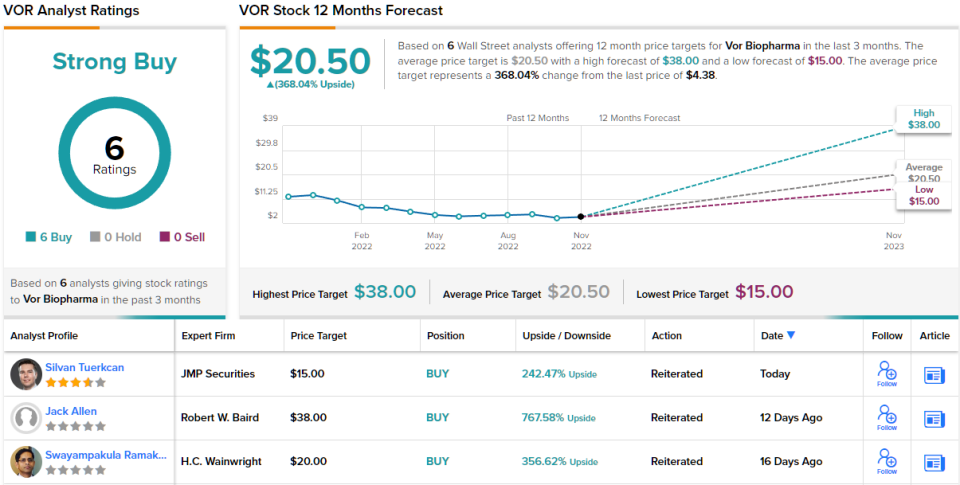
All of the above clearly shows why Tuerkcan is currently on the side of the bulls. The analyst rates VOR as Outperform (i.e. Buy), while his $15 price target implies a ~242% gain over the next year. (To see Tuerkcan’s achievements, click here)
So that’s JMP’s view, what do the rest of the Street think of Vor’s prospects? It’s all on board, as it happens. The stock has a Strong Buy consensus rating, based on 6 Consensus Buys. The VOR is selling for $4.38 and the average analyst’s target price is $20.50, for an impressive 368% gain. (View VOR stock forecast on TipRanks)
To find great ideas for trading penny stocks at attractive valuations, visit TipRanks’ Best stocks to buyone tool that unifies all of TipRanks’ equity insights.
deny the responsibility: The opinions expressed in this article are those of prominent analysts only. Content is used for informational purposes only. It is very important that you do your own analysis before making any investment.